Catalogue
Mouse anti Human Matrix Metalloproteinase 3 (MMP3)
Catalog number: X2055M$304.00
Add To Cart| Clone | 1B4 |
| Isotype | IgG1 |
| Product Type |
Monoclonal Antibody |
| Units | 200 µg |
| Host | Mouse |
| Species Reactivity |
Human |
| Application |
ELISA Immunohistochemistry Western Blotting |
Background
The matrix metalloproteinases (MMP) are a family of peptidase enzymes responsible for the degradation of extracellular matrix components, including Collagen, gelatin, Fibronectin, Laminin and proteoglycan. Transcription of MMP genes is differentially activated by phorbol ester, lipopolysaccharide (LPS) or staphylococcal enterotoxin B (SEB). MMP catalysis requires both calcium and zinc. MMP-3, MMP-10 and MMP-11 (also designated stromelysin-1, 2 and 3, respectively) activate procollagenase. MMP-3 activation of procollagenase can occur via two pathways. Direct activation by MMP-3 is slow and activation by MMP-3 in conjunction with tissue or plasma proteinases is rapid. MMP-10 is expressed in small intestine, and at lower levels in lung and heart. MMP-11 is specifically expressed in stromal cells of breast carcinomas and contributes to epithelial cell malignancies.
Synonyms: SL-1, Matrix metalloproteinase-3, MMP-3, Transin-1
Source
Immunogen: Hybridoma produced by the fusion of splenocytes from BALB/c mice immunized with a synthetic peptide derived from the C-terminus of the human MMP3 protein and mouse myeloma Ag8563 cells.
Product
Product Form: Unconjugated
Formulation: Provided as solution in phosphate buffered saline with 0.08% sodium azide
Purification Method: Protein A/G Chromatography
Concentration: See vial for concentration
Applications
Antibody can be used for Western blotting (1-2 µg/ml) and immunohistochemistry on formlin-fixed paraffin-embedded tissue sections (1-5 µg/ml). Optimal concentration should be evaluated by serial dilutions.
Functional Analysis: Western Blotting
Positive Control: Oesophageal and gastric tissues. Expressed at higher levels in tumor.
Storage
Product should be stored at -20°C. Aliquot to avoid freeze/thaw cycles
Product Stability: See expiration date on vial
Shipping Conditions: Ship at ambient temperature, freeze upon arrival
Caution
This product is intended FOR RESEARCH USE ONLY, and FOR TESTS IN VITRO, not for use in diagnostic or therapeutic procedures involving humans or animals. It may contain hazardous ingredients. Please refer to the Safety Data Sheets (SDS) for additional information and proper handling procedures. Dispose product remainders according to local regulations.This datasheet is as accurate as reasonably achievable, but our company accepts no liability for any inaccuracies or omissions in this information.
References
1. Knauper, V., et al. (1996). Activation of human neutrophil procollagenase by stromelysin 2. Eur. J. Biochem. 235(1-2);187-191
2. Saus, J. et al. (1988). The complete primary structure of human matrix metalloproteinase-3. Identity with stromelysin. J. Biol. Chem. 263;6742-6745
Protein Reference(s)
Database Name: UniProt
Accession Number: P08254 (Human)
Species Accession: Human
Safety Datasheet(s) for this product:
| EA_Sodium Azide |
|
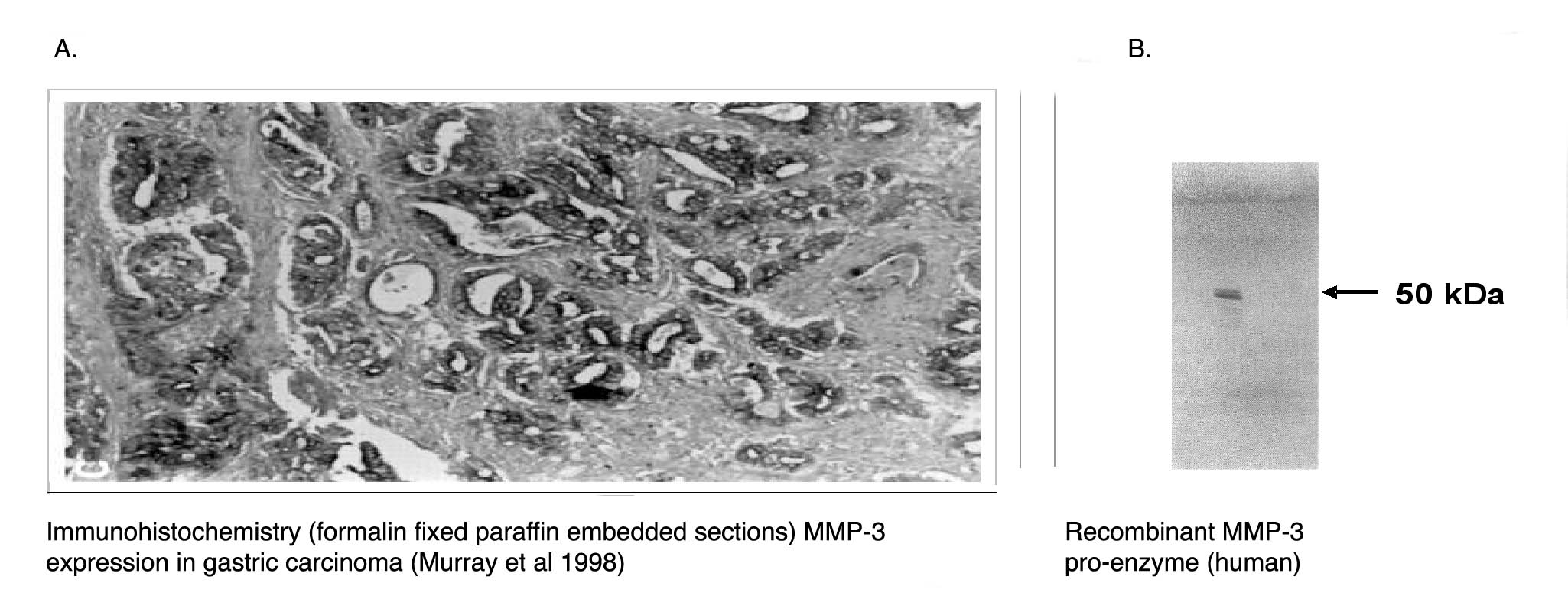
" Left: Immunohistochemical staining of human gastric carcinoma tissue using MMP-3 antibody (Cat. No. X2055M). Right: Western blot of recombinant human MMP-3 proenzyme using MMP-3 antibody." |

" Left: Immunohistochemical staining of human gastric carcinoma tissue using MMP-3 antibody (Cat. No. X2055M). Right: Western blot of recombinant human MMP-3 proenzyme using MMP-3 antibody."
